|
■
Home ■ site map |
|||||||||||||||||||||||||||||||||
|
BLOG: June 2010 - December 2013 III - ALTERNATIVE BREAST CANCER SCREENING TESTS 2. Better X-ray alternatives to mammography?
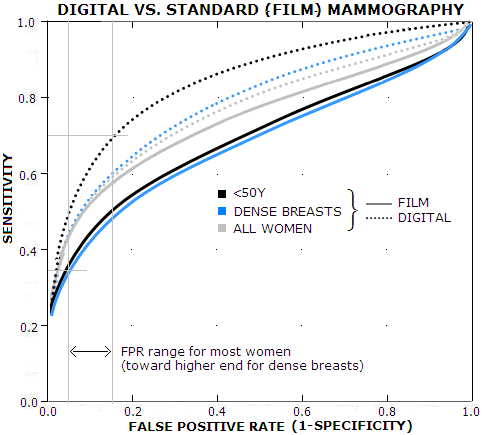
As the standard X-ray mammography, either film or digitalized, rapidly loses its cult-like status as the test of choice for periodic screening for breast cancer, alternative tests are quickly coming into focus. For now, all the alternatives seem to be presented mainly as complementary tools to remedy significant shortcomings of the conventional X-ray mammography. But it won't take long before a new test, or tests, will take its place. Alternative tests using different kind of ionizing radiation, gamma rays, as well as those not using ionizing radiation are addressed elsewhere. Here, we'll take a closer look of the alternative breast imaging technologies that do use X-rays, but in some ways are superior to the standard X-ray mammography. What is common to these new breast imaging techniques is that, unlike the standard X-ray mammography, which uses film to create the image of irradiated breast, they use digital detectors coupled with computer software. However, other recently developed tests, for instance, ultrasound, also use digitalized, computerized technology, thus the term "digital mammography" does not have specific meaning: it can refer to at least several different breast imaging techniques (i.e. mammography), including standard digitalized X-ray mammography, modalities of breast ultrasound, dedicated breast CT, breast tomosynthesis, and some others. For that reason, the term "digital mammography" is not used for digitalized standard mammography, as it is commonly the practice. This particular breast imaging technique was made so dominant in the medical diagnostic field and in public, that it has taken monopoly over the "mammography" term, which does not belong to it. Digitalized X-ray mammography The first alternative to the standard (film) X-ray mammography that came along was its own digitalized and computerized version. Despite the digital unit being about four times more expensive, it is quickly replacing the older version. According to most recent estimates, about 2/3 of all standard film mammography units in the U.S. have been replaced with digitalized units. Despite some articles making the digitalized version look like a big step ahead vs. film version (something should justify that huge extra spending of taxpayers' money), the consensus seems to be that there is no significant advantage of digitalized vs. non-digitalized standard mammography with respect to screening efficacy. Studies mainly indicate similar performance level for the two, with one study indicating significantly improved accuracy of digitalized X-ray mammography for women subpopulations below age 50 (22% larger area under the specificity/sensitivity, or ROC curve), for premenopausal and perimenopausal (22%), as well as those with dense breasts (15%, Diagnostic Performance of Digital versus Film Mammography for Breast-Cancer Screening, Pisano et al. 2005, for 42,700 asymptomatic women screened at 33 sites in U.S. and Canada). Following graphs (receiver operator curves, ROC) replicates performance plots presented in this study, for three population subgroups: under 50y of age, women with dense breasts and all women (mean age ~55y, approximately 40-70y range).
However, since this same study had also found nearly identical accuracy for the two imaging technologies - film and digital mammography - in the entire study population, this directly implies somewhat inferior accuracy of digitalized X-ray mammography in the remainder of the population (i.e. women over 50, postmenopausal and women with normal-to-low breast density, i.e. fatty breasts). Not unusual in this field, study results are not making much sense, with no rational explanation for digital mammography, relying on that same x-ray image smeared by the overlapping dense tissues of the breast, to have significantly higher sensitivity for women below 50y than for all-women group, or women with dense breasts. In another study, non-digital standard mammography outperformed the digital by 22% in cancer detection rate, which hasn't been statistically significant (Lewin et al. 2002). Research on this particular modification of the standard mammography is still scarce, but there is no reason to expect major changes in its efficacy: it is still based on that same shadowy 2-D image. Study result, mentioned above, indicating higher sensitivity and specificity doesn't seem logical, since the increased test sensitivity (i.e. less false negatives) inevitably comes at a price of lowered specificity (more false positives). Also, since at present we are not able to tell with certainty which abnormal tissue growth will progress into breast malignancy, and which will not, higher test sensitivity also inevitably increases the rate of overdiagnosis and overtreatment. The advantage of the digitalized conventional mammography vs. non-digital is in easier data storage, retrieval and transport. Also, direct access to the image during the session allows for more efficient use of radiation, hence for generally somewhat lower radiation exposure as well. But none of it changes significantly the overall questionable positives-to-negatives ratio of the standard film variety. Dedicated breast CT It is known that the computed tomography (CT), diagnostic imaging technique in which a 3-D image of the body area of interest is constructed by use of a computer algorithm from a series of 2-D images taken at different angles, offers generally better quality and higher detection level than X-ray mammography. However, its two main drawbacks - high cost and high radiation exposure relative to the standard X-ray mammography - were both, limiting its use for breast diagnostics and practically excluding it as a possible test for periodic breast screening. This might be changing, and rather quickly. Reconfiguring CT scan for breast imaging - into so called dedicated breast CT - nearly eliminated both drawbacks, at least with respect to the standard X-ray mammography. Instead of the full 360-degree rotation of the X-ray emitter around chest cavity, dedicated breast CD uses much smaller unit that rotates around breast protruding from an opening on a radiation shielded horizontal table. This re-arrangement not only spares chest cavity from exposure to radiation, which is significant drawback of the standard CT scanner when imaging the breasts, it also allows for much lower total radiation level in obtaining high resolution images. Breast CT radiation dose, according to Boone et al., is at the same level with that delivered by 2-view mammography for given breast compressed thickness of about 5cm (in the standard mammography; as mentioned before, there is no breast compression with the breast CT), exceeding somewhat the conventional mammography dose with smaller breasts, but delivering less radiation with larger breast.
On the average, radiation risk with breast CT is similar to that
with This means that breast CT delivered radiation should be similar to that of X-ray mammography for medium and thin breasts, and lower for those thicker than average. Unlike X-ray mammography, breast density does not present an obstacle to the test accuracy. High signal-to-noise ratio of the test images enables detection of significantly smaller tumors, down to 2-3mm, up to several times smaller than with the standard X-ray mammography (Dedicated breast CT: Radiation dose and image quality evaluation, Boone et al. 2001). Taking an average 100 days as the period of time needed for the tumor cells to double in number, (i.e. 400 days for doubling of the tumor diameter), this translates in up to several years earlier detection than with the standard mammography, at least in theory. Why? Even if breast CT turns out to have significantly superior detection level, that is not necessarily an advantage. Better sensitivity in general reduces the number of false negatives, but at a price of the higher rate of false positives, overdiagnosis and overtreatment. Only if the former is more significant than the latter, higher detection rate would count as advantage, but that is highly unlikely. Standard mammography already detects significantly more abnormal growths in the initial stage, but it did not translate into BC mortality reduction that would even offset all the negatives. The only limitation of the dedicated breast CT was its inferior spatial resolution. Despite more contrasty images, its resolution of small details - specifically, microcalcifications - was initially inferior to that of the standard mammography. That, however, may not be a lasting limitations, as it was reported recently that this limitation can be overcome with a modified technology, which would make breast CT superior in all aspects of imaging, while keeping radiation dose comparable to that of the standard mammography (Concepts for High-Resolution CT of the Breast, W. Kalender 2010). The absence of breast compression - a truly sore (and possibly dangerous) point of X-ray mammography - certainly adds to the positives of this imaging technique. According to Boone, breast CT would cost about twice more than the standard digital mammography unit. It is uncertain how much would it cost with the Kalender's improved technology. Considering that the mere improvement in test detection capability does not solve the main problems of the standard mammography screening - false positives, overdiagnosis and overtreatment - and that early detection itself did not show convincingly that it reduces BC mortality, it is not apparent why doubling the expense without having these issues resolved would be worth it. Breast tomosynthesis Breast tomosynthesis is similar to the dedicated breast computed tomography (CT) in that it uses computer based reconstruction of a 3-D image from a series of 2-D X-ray images taken at different angles. Unlike CT, the X-ray unit completes only a fraction of full circle around target tissue, i.e. breast - anywhere from 15 to 60 degrees arc - hence also uses different image-constructing algorithm. Due to the relatively small angle, similarly to the standard X-ray mammography, it may require two views, medio-lateral oblique (abbr. MLO, side view), and craniocaudal (abbr. CC, top-to-bottom). Breast in tomosynthesis needs to be positioned and immobilized same as with X-ray mammography but, according to the manufacturer, requires less pressure. Due to its different methods of image acquisition and algorithms vs. breast CT, imaging quality in breast tomosynthesis is also somewhat different. The latter is expected to have somewhat lower contrast resolution (i.e. resolution of low-contrast details), but higher spatial resolution (limiting resolution for contrasty details). With respect to. X-ray mammography, breast tomosynthesis is expected to have better contrast resolution, and somewhat inferior spatial resolution, but not as much as breast CT. That places it between breast CT and standard X-ray mammography with respect to the image quality, one of the main determinants of test reliability. That said, there are good and bad sides to better resolution. The good side is that it is likely to increase test sensitivity, i.e. ability to detect breast cancer when it is present (in other words, less false negatives). On the other hand, that will likely lower test specificity, i.e. efficiency in furnishing a negative finding when cancer is not present (more false positives). Neither breast CT nor breast tomography - or, for that matter, standard X-ray mammography - have the advantage of detecting mechanical/dynamic tissue properties that some other tests - like ultrasound - have. And neither puts the radiation risk away. Breast tomosynthesis uses similar X-ray strength (peak kilovoltage 25-35kV) as the standard mammography, thus the dose is directly comparable. The 3-D image is constructed of a series of quick (5-10 seconds total) low-dose exposures, each about 10%, or so, of the average single-view standard mammogram. Since exposures are taken at every 1-2 degrees, with, a 20-degree imaging angle would consists of up to 20 exposures with up to double the single-view radiation of the standard mammography. The problem is that it hasn't been determined yet what specific angle width and imaging frequency would be accepted as standard, so the radiation dose delivered by this test remains uncertain. The exact radiation dose here is also uncertain for another reason. Equipment manufacturers (GE Healthcare, Siemens Medical Solutions and Hologic, Inc.) maintain that radiation dose per view will be identical to that in the stsndard X-ray mammography. The problem here is that it is still undetermined how many views - and how many projections per view, as mentioned above - will be necessary with the breast CT to achieve needed efficacy.
More likely than not, a single view won't be enough, and with
two views the
radiation dose would be similar to that with conventional
But no one can really tell for sure, at this point, what dose of radiation per view tomosynthesis will need to produce competitive - or better - image quality. The manufacturers and promoters seem to be basing their equal-dose (vs. standard mammography) assessment on the relatively small scan arc (15-30 degrees) and low to medium number of projections (7-15). On the other hand, a recent small study, cited for its results supportive of tomosynthesis' efficacy, used 50-degree arc and 25 projections per view, with the radiation dose per view double that of the standard mammography (Lo et al. 2007, for Siemens Medical Solutions). It used two views for diagnostic cases, and a single view for screening. In all, it seems more likely than not that tomosynthesis will deliver somewhat higher radiation dose than the conventional X-ray mammography. The bottom line is that neither breast CT nor tomosynthesis would reduce the radiation risk with respect to the current screening test. Also, both are more expensive, with tomosynthesis having the advantage of relatively low cost if installed with an existing digital X-ray mammography equipment (considering that most of centers have such equipment at present, it is a significant advantage cost-wise). So it all boils down to their detection efficacy; if their seeming superiority to conventional X-ray mammography in both, sensitivity (i.e. lower false negatives) and specificity (lower false positives) is confirmed to be substantial, it might justify their higher cost. But, as already mentioned, it is more likely that higher sensitivity would result in more false positives and, considering our inability to tell which abnormal growth will become symptomatic, more overdiagnosis and overtreatment. The clearer image could somewhat reduce the rate of biopsies, and the 3-D quality would be helpful in (biopsy) sample localization. But that alone wouldn't be enough. For the final conclusion, we'll have to wait for their more widespread use, if it ever takes place. One thing is certain:
it is not a good option to have them as an
add-on to the the scenario most often mentioned at this moment. Even if it would somewhat increase the overall efficacy of screening (according to some early studies, by 30-40%), it would come at the cost of significantly higher radiation exposure. And, all indications are that radiation exposure from the conventional mammography alone is already unacceptably high for radiation-sensitive individuals. Standard CT scan Standard CT scan for breast imaging also uses X-rays, but it delivers radiation dose comparable to that used for chest CT, which is a dozen or more times higher than with the conventional mammography. In addition to the breast tissue, other organs in the chest cavity are also irradiated, broadening the risk. Since the scanner makes a full circle around the chest, cardiac and respiratory tissue, and its motion, get in the way, lowering image contrast and resolution. Adding to it high cost makes this breast examination method even less suitable for any form of periodic screening. Similarly to these recent X-ray technology modifications, there are new developments focused on breast cancer detection in the area of nuclear medicine, using gamma-radiation. More on the next page. YOUR BODY ┆ HEALTH RECIPE ┆ NUTRITION ┆ TOXINS ┆ SYMPTOMS |
|||||||||||||||||||||||||||||||||